Facilities

Highlights
- Exercise and Rehabilitation Training Facilities
- Large Conference Room / Lecture Area
- Offices for Faculty, Staff, and Research assistants
- Secure Data Storage Area
- 12 Bed Facility for Overnight Research Studies
- Metabolic Kitchen
- DEXA Body Composition and Bone Density
- Resting Energy Expenditure Rooms
- Cardiopulmonary Exercise Testing
- Isotonic and Isometric Strength Assessment
- Anaerobic Capacity Testing
- Balance Assessment
- Medical Exam & Procedure Rooms
- Cognitive and Neuromuscular Assessment
- Comprehensive Wet Lab Facilities
- Short-Radius Human Centrifuge
Overview
The Human Clinical Research Building (HCRF) is a 23,000-square-foot biomedical research facility specifically designed to perform human clinical trials at TAMU. Built in 2017, this modern building contains hospital-style research rooms with 12 beds with adjacent bathrooms and showers for overnight stay research; a metabolic kitchen to prepare and administer meals; a nursing station that can monitor and communicate with participants in study rooms; a sample processing lab; a comprehensive physiological assessment lab with 6 exam rooms that can conduct a wide variety of cardiovascular, metabolic, exercise, body composition, balance, and biomedical assessments; a comprehensive wet lab to analyze blood, urine, feces, and/or tissue samples; a clean room for mixing pharmaceutical grade oral and intravenous solutions; a comprehensive rehabilitation and exercise training facility with an exam/phlebotomy room and bathroom/changing area; a short arm human centrifuge for gravity dose research (which will support spaceflight research as well as collaboration with NIH); a computerized-access data storage area; a conference/lecture room; a break room; and, offices for up to 24 faculty/staff and 24 research assistants and students.
Facility Management
From 2017 – 2022, the HCRF was managed by the School of Education and Human Development (SEHD) Research and Development Office under the direction of Dr. Richard Kreider (Executive Director) and Nicolaas Deutz, MD, PhD (Clinical Research Director). This included development of the Human Clinical Core service center that offers a number of clinical research services on a fee-for-service (FFS) basis for TAMU faculty and sponsors. In September, 2022, as part of the University’s new Path Forward realignment, the HCRF was transferred to TAMU Health under the leadership of Dr. Jon Mogford, COO and Sr. VP. The HCRF facility remains FFS with Dr. Kreider, on the faculty of the SEHD, directing the on-going service program supporting several external and internal research projects. The new alignment with the TAMU Health Science Center provides greater opportunity to provide medical, nursing, and rehabilitation service support to the HCRF. The TAMU-HSC is composed of five schools: Dentistry, Medicine, Nursing, Pharmacy, and Public Health. Expanding and growing the utilization of the HCRF collaboratively within our Texas A&M University and System is a top priority. Further growing partnerships across the state and nation to advance science and health is a strategic goal. As part of this expanded effort, effective January 1, 2024, the HCRF will be housed under the direction of the Huffines Institute for Sports Medicine and Human Performance. This will expand university multidisciplinary efforts to conduct human performance and clinical research. The Aerospace Short Arm Centrifuge is administratively managed and directed by the College of Engineering in collaboration with the TAMU HSC, where it is included in the FFS catalog of offered services at the HCRF.
Clinical Research Unit
The HCRF currently contains seven hospital-style study rooms that contain 12 beds for overnight research. One room has four beds, two rooms have two beds, and four rooms have single beds. This provides the flexibility to keep participants together or isolate them for various procedures.
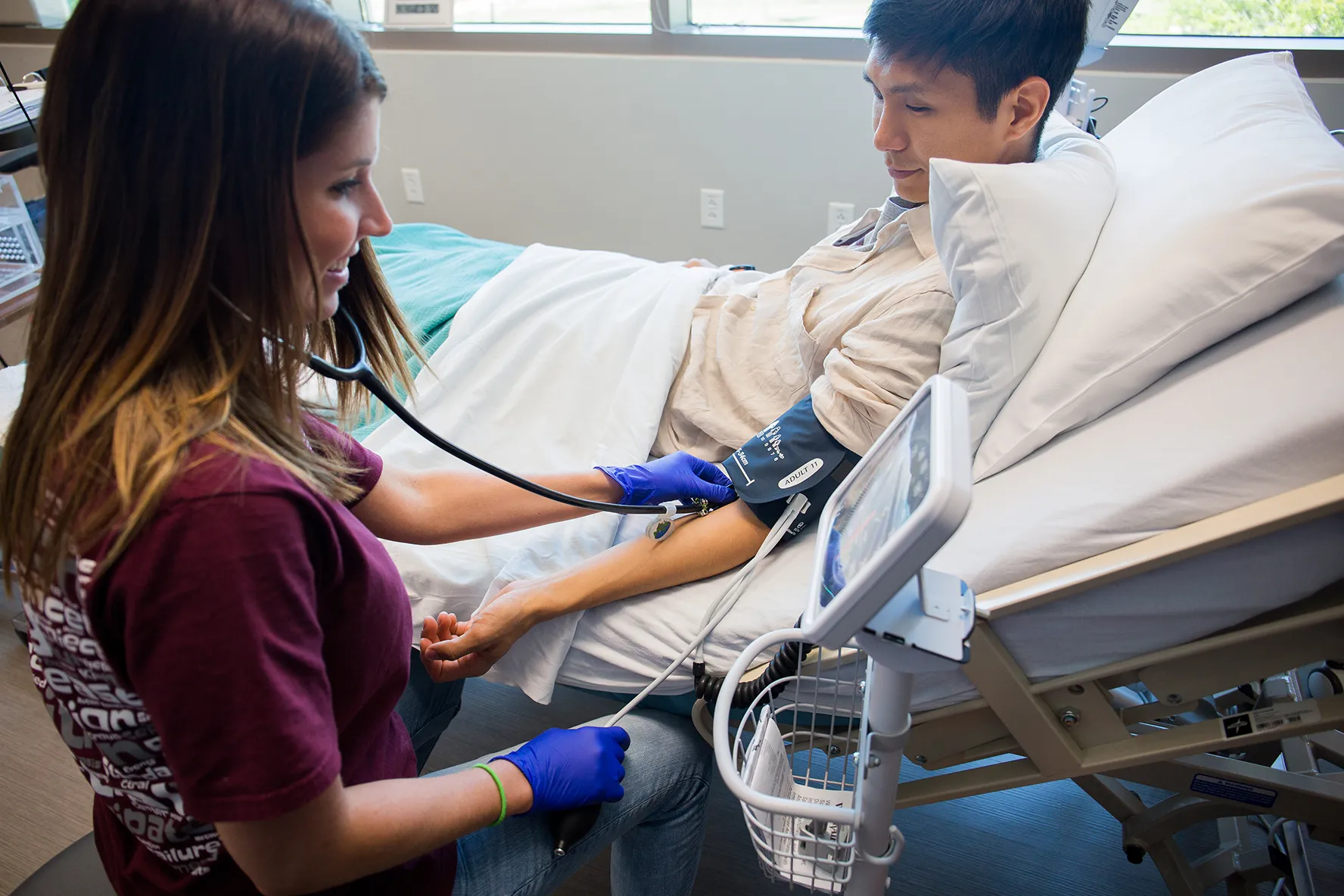
Each study room contains at least one hospital grade adjustable and moveable bed with access to oxygen; electrical and Internet lines outlets for monitoring equipment; hemodynamic and vital sign monitoring devices; video monitoring and audio communication capabilities; a television for personal entertainment; a moveable and adjustable phlebotomy chair; clinical space (storage cabinets, sinks, etc.); and adjacent private bathrooms and showers. Two study rooms have large bariatric beds and bathrooms/showers for handicap and wheelchair access.
The HCRF was designed with wide doors, hallways, and an elevator capable of moving moveable beds and phlebotomy chairs to physiological assessment in the Exercise & Sport Nutrition Lab (ESNL) or downstairs to rehabilitation/training and/or the SAC. Most of the physiological testing equipment needed is also portable and can be moved into study rooms to do assessments bedside.
The large study room with four beds is primarily used for Center for Translational Research in Longevity (CTRAL) funded research projects. They also use some of the other study rooms for individual assessments (e.g., pulmonary assessment, nap studies using electroencephalogram (EEG) analysis, neurological testing, etc.). Depending on how many beds are needed for bed rest studies, some of these assessments could be performed in other locations (e.g., exam rooms in 206). We currently have four Lunar beds with head-down tilt (HDT) capabilities that we obtained from NASA/UTMB in storage that can be put in these rooms, as needed. Depending on how many beds are required for HDT bed rest studies, we can replace other standard hospital beds with HDT beds. If more beds are needed, we could do some renovations to expand the study room space. Additionally, the HCRF was designed to be able to add a third floor. A long-term investment would be to add more clinical study rooms/beds in space that can be developed on the third floor of the building if needed. Options for expansion are further discussed in section.

Participant Monitoring
There is a nursing/participant monitoring station outside of the study rooms in the clinical research unit. This includes a computer-based monitoring system that research personnel can video, record, and communicate with participants as well as monitor participant vital signs. The HCRF has a Medical Director (J.P. Bramhall, MD) who reviews all IRB applications. CTRAL also employs at least one research nurse to oversee clinical research protocols with older and clinical populations.

To do bed rest studies, we hire additional physicians, nurses, and orderly staff as needed to monitor and take care of research participants on a 24-hour basis. The HCRF partners with TAM Health, the College of Medicine, and School of Nursing that helps provide staff, residents, doctoral students, and interns to support clinical testing as needed.
We also have a number of exercise physiologists and dietitians (typically staff or PhD students) to conduct Pre- and Post-Bed rest study measurements and rehabilitation sessions.
Finally, Dr. Sowinski is a trained chef and coordinates meal preparation in the metabolic kitchen. Depending on how much bloodwork can be done at commercial labs, we will also employ biochemistry technicians as needed to analyze samples.
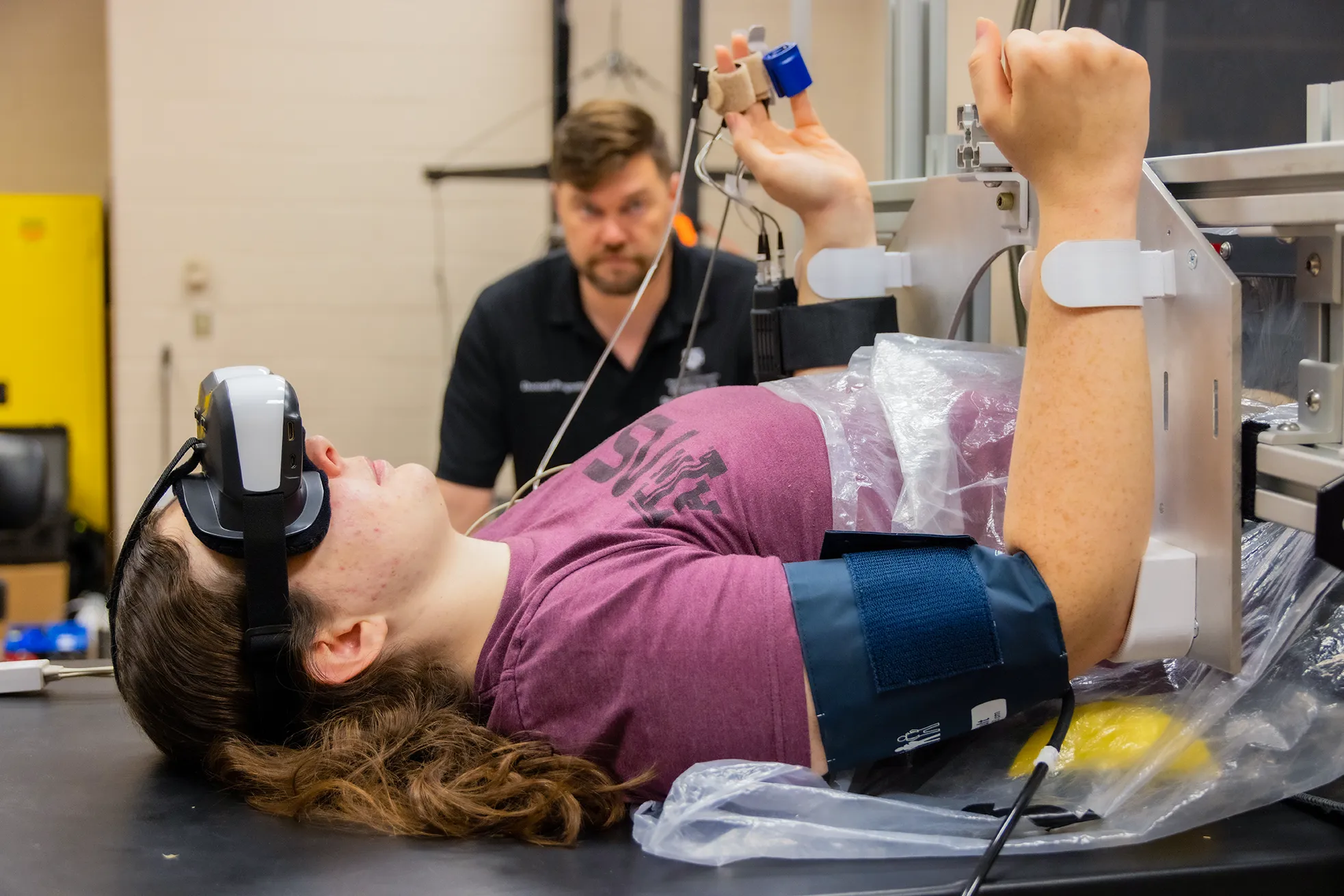
Assessments
The HCRF contains a wide array of physiological and medical monitoring systems (see Services and Research Capabilities). This includes the ability to collect, process, and store blood, urine, feces, and tissue samples as well as conduct various clinical assessments (e.g., body weight, height, resting blood pressure and 12-lead ECG tracings, resting energy expenditure (REE), energy intake analysis, multi-frequency bioelectrical impedance (BIA) tests, dual-energy x-ray absorptiometry (DXA) scans, cardiopulmonary exercise tests with CO2 rebreathing determined cardiac output measures, ultrasound heart and venodilation studies, fNMR scans, MRI, isokinetic and isotonic strength tests, anaerobic capacity tests, cognitive function tests, nutrition analysis, psychological testing, etc.).
Most physiological testing is conducted in ESNL that is in room 206 next to the clinical research unit. However, most of the equipment needed to do these tests is portable so we can do a number of in-bed assessments (e.g., blood, urine, feces collection, weight, heart rate, blood pressure, ECG, REE, EEG, BIA, supine cycling and/or arm ergometer exercise training and cardiopulmonary tests, ultrasound, fNMR, cognitive function tests, psychological inventories, etc.). We can also train medical support staff (i.e., nurses, residents, orderly’s) to assist in collecting some of this data as needed.
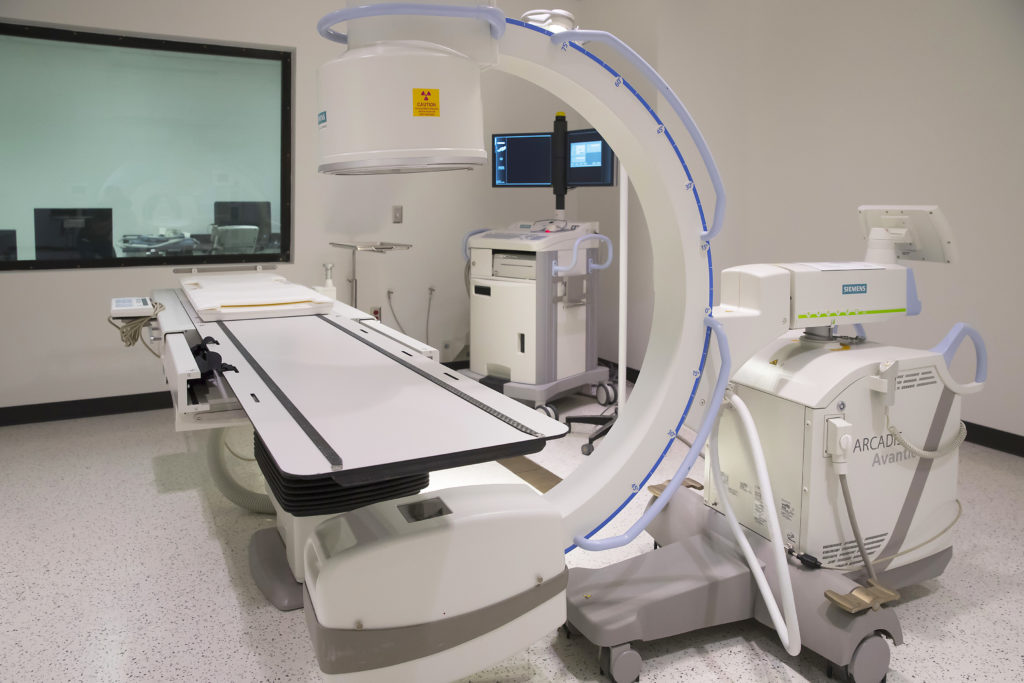
Imaging Capabilities
MRI imaging capabilities in the Texas A&M Institute for Pre-Clinical Studies (TIPS) which is about half mile away from the HCRF. Participants are schedule for appointments as needed. If needed, we can transport participants in a modified gurney for imaging (including for strict head-down tilt studies). However, we are also exploring obtaining the Swoop Portable MRI Imaging System that can do scans in the study rooms while participants are in their bed.

Diagnostic Capabilities
In terms of laboratory services, we have experience collecting blood, urine, feces, and tissue samples using fine needle aspiration or percutaneous needle biopsy techniques. We currently have eight monitored ultralow freezers to store samples. The wet lab in the HCRF contains analyzers capable of running general clinical markers (e.g., cell blood counts, serum clinical markers, etc.) and specialty assays (e.g., spectrophotometers, plate readers for ELISA and EIA analysis, Luminex multiplex system, HPLC, aggregometer), cell culture capabilities, and LC-MS/MS, and GC-MS/MS systems for analysis of small metabolite and stable isotope analysis.
The latter capabilities provide a unique opportunity to do detailed metabolic studies to understand how bed rest impacts energy expenditure, metabolism, and muscle wasting. We typically send samples to external CLIA certified clinical labs (e.g., Quest, LabCorp, Clinical Lab Pathologies) for general whole blood and serum clinical chemistry analysis. We do platelet aggregation studies after sample collection and store samples to batch analysis of hormones, cytokines, inflammatory markers, immune markers, and isotope analyses. We also have experience collecting feces for microbiome analysis.
The HCRF serves as the location for our Human Clinical Core fee-for-service program. We also collaborate with several other centers and core facilities on campus for additional analyses (e.g., genetic analyses, flow cytometry, vitamin, and mineral status, etc.). Therefore, the HCRF contains and/or has access to a wide array of medical capabilities and diagnostic lab services to support Bed rest studies

Exercise Training & Rehabilitation
The HCRF contains a comprehensive training facility to do exercise and rehabilitation intervention studies in healthy and special populations. The rehabilitation area has oxygen availability, physical therapy equipment (i.e., stretching bed, rehabilitation bands and light weights, isokinetic dynamometer), cardiovascular training equipment (e.g., treadmills, bicycle ergometer, recumbent bicycle ergometer, arm ergometer), and resistance training equipment (e.g., Keiser computerized equipment, Curves hydraulic equipment).
We use this equipment primarily for rehabilitation of individuals with medical conditions in which exercise may benefit, cardiopulmonary rehabilitation, rehabilitation from bed rest studies, and elderly. Additionally, we have an exercise training facility for healthy and advanced training equipped with resistance exercise machines, free weights, dumbbells, treadmills, and bicycle ergometers. We use this equipment in exercise and nutritional intervention studies. We will need this equipment for participants before starting bed rest studies and to rehabilitate participants after long-term bed rest studies.

Aerospace Engineering SAC Centrifuge
Construction has been completed on an Aerospace Research Annex to the HCRF that houses the short-arm centrifuge (SAC) formerly built by NASA and housed at the UTMB in Galveston, Texas. This is the only short-arm human centrifuge (SAC) of its kind in the United States and has been used for NASA peer reviewed research to understand the effects of gravity dose on human physiology, and to prepare for human exploration in reduced gravity environments of the Moon and Mars.
Retired astronaut and Professor of Aerospace Engineering, Dr. Bonnie J. Dunbar, obtained approval to acquire the SAC from NASA JSC through the Stevenson Wydler Technology Innovation Act, and it is now owned by TAMU TEES.
After considering various location options on the TAMU campus, she decided that the HCRF would be an ideal location because of the ability to provide physiological and clinical assessment support and the potential to perform bed rest studies that would involve use of the SAC. Dr. Dunbar invested start-up funds (Chancellors Research Initiative, CRI) of about $1.2 million as well as $0.66 million of the TAMU Vice President of Research (VPR) Research Development Fund (RDF) grant to support this effort.
The human centrifuge facility is directed by Dr. Dunbar and Dr. Ana Diaz Artiles, who performed research using a similar facility when she was at Massachusetts Institute of Technology. Dr. Diaz Artiles and Dr. Dunbar currently have two NASA HRP funded grants which use the Centrifuge to generate gravity dose data. The Human Centrifuge is currently under final integration by KBR, the contractor who design the system. KBR is also funded to provide significant upgrades to the control room and instrumentation.
